A NEW Covid jab for Britain is effective in 89.3 per cent of cases, it was revealed last night.
Sixty million doses of the Novavax vaccine will be produced on Teesside — as the EU is trying to scupper exports of jabs.
😷 Read our coronavirus live blog for the latest news & updates
The drug, shown in UK trials to be effective against the Kent variant of Covid, was hailed by PM Boris Johnson.
Britain's incredible vaccine campaign got a triple boost with the news of 150million extra jabs.
The Government has secured 60million doses of the drug — which experts said was also 85.6 per cent effective against the sweeping UK variant of Covid-19.
The Sun can reveal results of trials for the one-shot US Janssen vaccine are expected imminently — with Britain having ordered 30million doses.
A French firm is using a Scottish factory to produce another 60million doses of a promising vaccine.
It means 120million of the extra jabs will be UK-produced.
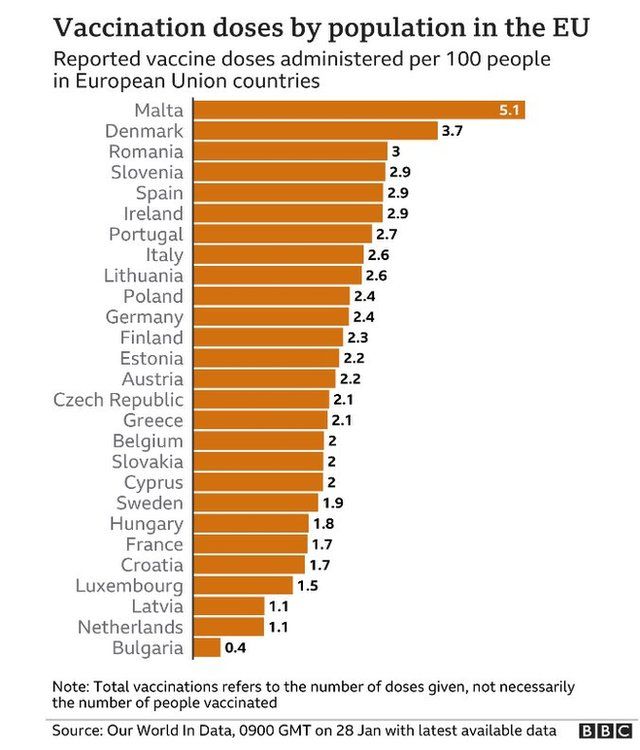
It came as figures showed 7,447,199 Brits have now received their first does, and 476,298 have had their second.
The Novavax jab’s 89.3 per cent efficacy in UK trials was called “spectacular” by expert Clive Dix.
It is likely to be approved next month and will be manufactured in Stockton-on-Tees for use this summer. The factory, owned by Fujifilm, has capacity to ramp up production to 180million doses.
Novavax boss Stanley Erck said it has been shown to work against “UK and South Africa variants”.
Vaccine Minister Nadhim Zahawi, one of 15,000 UK volunteers to take part in the Novavax trials, said: “I am particularly thrilled to see such positive results.”
Health Secretary Matt Hancock said: “If approved by the regulator, the vaccine will be a significant boost and another weapon in our arsenal to beat this awful virus.
'UK AT THE FOREFRONT OF ANOTHER MEDICAL BREAKTHROUGH'
“I’m proud the UK is at the forefront of another medical breakthrough and I want to thank the brilliant scientists and researchers, as well as the tens of thousands of selfless volunteers who took park in clinical trials.
“The NHS stands ready to roll this vaccine out as quickly as possible if it is authorised.”
PM Boris Johnson said it was “good news”.
Meanwhile French firm Valneva has started large-scale manufacturing of its vaccine in Livingston, West Lothian.
It can supply up to 250million shots annually. On top of this, US tests on the Janssen vaccine are expected to pave the way for 30million doses to start arriving in the UK as early as July.
Janssen is the Belgium-based vaccine arm of US pharmaceutical giant Johnson & Johnson.
Through its Vaccines Taskforce, the UK has now secured early access to 367million jabs.
There was further good news when Israel announced that less than 0.04 per cent of patients who received two doses of the Pfizer vaccine have developed Covid.
But holidays to European hotspots will be off until they sort their shambolic vaccination rollout. Senior cabinet minister Michael Gove has hinted that stricter border measures could be in place for months, even after the UK comes out of lockdown.
Yesterday the UAE, a favourite of social media influencers, was added to 30 no-go countries.
An estimated 24,000 Brits across the globe now face dashing back to avoid having to quarantine in hotels on arrival, expected to be imposed on February 8. But for the next week, arrivals will have to isolate for ten days at home.
Mr Gove said: “I think it’s important to stress that people shouldn’t expect any premature lifting of these restrictions.”
GOT a story? RING The Sun on 0207 782 4104 or WHATSAPP on 07423720250 or EMAIL exclusive@the-sun.co.uk
https://news.google.com/__i/rss/rd/articles/CBMiR2h0dHBzOi8vd3d3LnRoZXN1bi5jby51ay9uZXdzLzEzODgyMTEyL25vdmF2YXgtdmFjY2luZS11ay1jb3ZpZC1zdHJhaW4v0gFLaHR0cHM6Ly93d3cudGhlc3VuLmNvLnVrL25ld3MvMTM4ODIxMTIvbm92YXZheC12YWNjaW5lLXVrLWNvdmlkLXN0cmFpbi9hbXAv?oc=5
2021-01-29 08:19:00Z
52781338842931