A fourth COVID-19 vaccine could be approved for use in the UK within weeks after late-stage trials suggested it was 89% effective in preventing coronavirus.
The prime minister has said the Novavax jab is now going to be assessed by the Medicines and Healthcare products Regulatory Agency (MHRA).
If approved, the vaccine would start to be rolled out in the second half of 2021. The UK has already ordered 60 million doses, which are going to be manufactured in Stockton-on-Tees.
More than 15,000 people aged 18 to 84 took part in the Phase 3 clinical trial in the UK - about a quarter of whom were over the age of 65.
The extensive study also showed that the Novavax jab offered 86% protection against the new British strain of COVID-19, which is up to 70% more transmissible.
A smaller, separate trial also showed that it was about 60% effective against the South African variant, despite concerns that this strain may not respond to vaccines.
Health Secretary Matt Hancock has said the NHS is prepared to start rolling out the vaccine quickly if authorised by the MHRA, adding: "This is positive news and, if approved by the medicines regulator, the Novavax vaccine will be a significant boost to our vaccination programme and another weapon in our arsenal to beat this awful virus."
Vaccines minister Nadhim Zahawi was among those who took part in the Novavax trial, and said he was "particularly thrilled" to see such positive results - thanking fellow volunteers for taking part.
The breakthrough comes as a row deepens between the EU and AstraZeneca over vaccine shortages in the trading bloc.
Despite Brussels calling for doses to be sent over from British plants to compensate for the shortfall, Cabinet Office minister Michael Gove has warned that the government won't allow vaccines intended for Britons to be sent to the EU.
Reports have suggested that the EU could block millions of coronavirus vaccines from being exported to the UK within days.
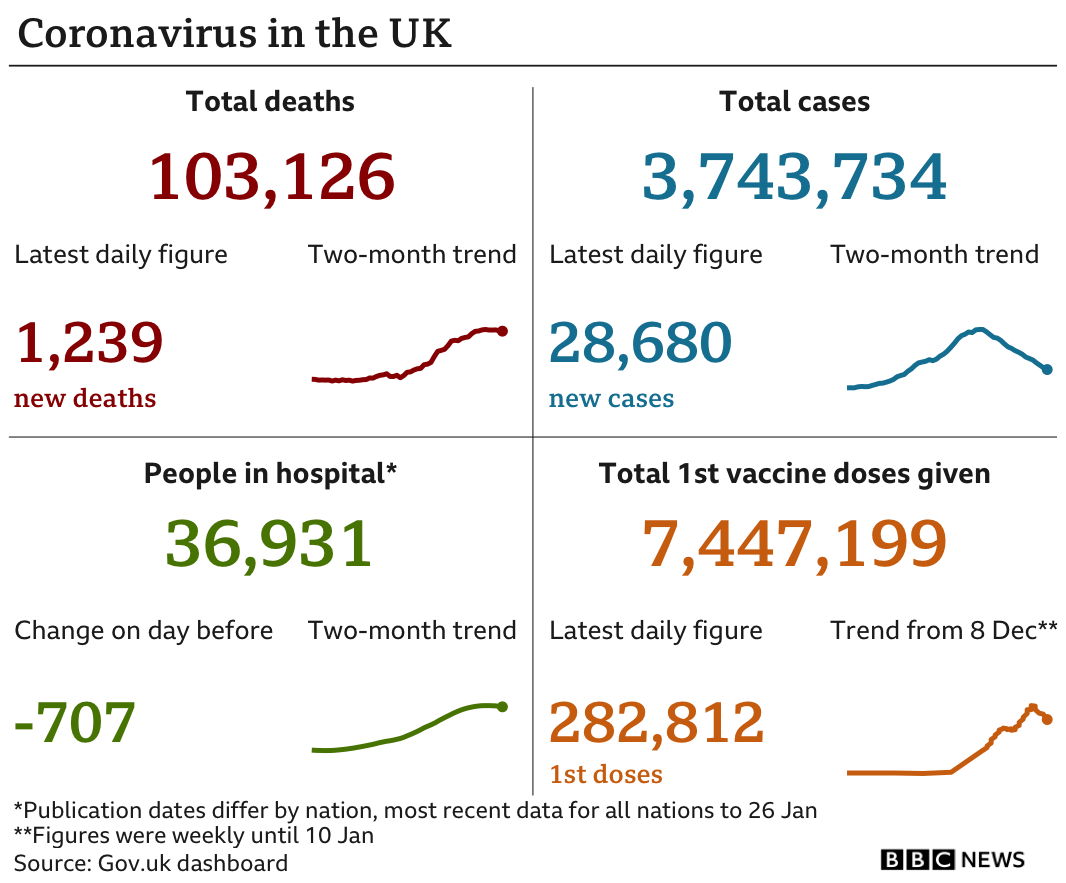
The latest figures show a further 1,239 people died within 28 days of testing positive for COVID-19, with another 28,680 lab-confirmed cases of coronavirus in the UK.
Novavax is currently stockpiling vaccines at six manufacturing locations - and is aiming for eight plants in seven countries to produce two billion doses a year.
The effectiveness of the Novavax jab is not far off the 95% success rate seen with the Pfizer/BioNTech and Moderna vaccines, which have already been approved for use, while the Oxford/AstraZeneca vaccine is up to 90% effective.
Dr Amesh Adalja, an infectious disease expert at the Johns Hopkins Centre for Health Security, said the 60% effectiveness against the South African variant of COVID-19 is still promising - not least because US regulators previously said they were prepared to approve a jab that was just 50% effective.
"We've gotten spoiled because we've seen the Moderna and Pfizer numbers," he said. "I know people are going to be alarmed, but 60% efficacy against the new variant is acceptable."
Subscribe to the All Out Politics podcast on Apple Podcasts, Google Podcasts, Spotify, Spreaker
Like the Oxford/AstraZeneca vaccine, the Novavax jab only needs to be kept at fridge temperature, meaning distribution and supply chain management is easier.
After an initial dose, a second shot is given three weeks later.
At a cost of $16 (£11.66) per vaccination, the Novavax jab is more expensive than AstraZeneca's, but cheaper than Pfizer and Moderna's offerings.
This sorely needed vaccine appears to be extremely robust
Analysis by Thomas Moore, science correspondent
The first of a second wave of vaccines is almost here.
This is a new kind of vaccine - instead of injecting the genetic material for the spike protein, this is the protein itself. Not the whole virus - it is just the protein which is injected, and it's got very, very good results.
It is 89% effective in the data that's came forward on Thursday night. And what's more, it worked really well in the people that it needs to work in - 27% of the people on the trial were over 65 and significantly, it worked against the new variants circulating here in the UK - there it was 86% effective.
In South Africa where there was another trial, again, a very worrying variant there - perhaps more worrying than the one here - but it was 60% effective there.
So it does seem to be an extremely robust vaccine.
Now it's a matter of getting it through the gates.
So you'd expect it to take about a month to get through the medical regulation, and then we can see 60 million doses coming our way.
And they will be needed, because at the moment we don't have enough vaccines for everybody who really needs it.
Over three nights, Sky News will host a series of special programmes examining the UK's response to the pandemic.
Watch COVID Crisis: Learning the Lessons at 8pm on 9, 10 and 11 February.
https://news.google.com/__i/rss/rd/articles/CBMiiAFodHRwczovL25ld3Muc2t5LmNvbS9zdG9yeS9jb3ZpZC0xOS1mb3VydGgtdmFjY2luZS1jb3VsZC1iZS1hcHByb3ZlZC1pbi13ZWVrcy1hcy10cmlhbC1zaG93cy1pdC1pcy1lZmZlY3RpdmUtYWdhaW5zdC11ay12YXJpYW50LTEyMjAxODMz0gGMAWh0dHBzOi8vbmV3cy5za3kuY29tL3N0b3J5L2FtcC9jb3ZpZC0xOS1mb3VydGgtdmFjY2luZS1jb3VsZC1iZS1hcHByb3ZlZC1pbi13ZWVrcy1hcy10cmlhbC1zaG93cy1pdC1pcy1lZmZlY3RpdmUtYWdhaW5zdC11ay12YXJpYW50LTEyMjAxODMz?oc=5
2021-01-29 02:00:55Z
52781337762950