The prime minister and Public Health England have both defended the use of the Oxford-AstraZeneca jab, after Germany recommended that it should only be given to people aged under 65.
Dr Mary Ramsay of PHE said the jab offers "high levels of protection" against Covid-19, particularly against severe illness.
Boris Johnson said he was not concerned by Germany's recommendation.
AstraZeneca said the jab's trial data supported efficacy in the over-65s.
Germany's vaccine committee said the AstraZeneca vaccine should only be given to people aged under 65, citing a lack of sufficient data to recommend use among older age groups.
The European Medicines Agency (EMA) is to decide on Friday whether to approve the vaccine for use across the EU.
But Dr Ramsay, head of immunisations at Public Health England, said: "Both the AstraZeneca and Pfizer-BioNTech vaccines are safe and provide high levels of protection against Covid-19, particularly against severe disease.
"There were too few cases in older people in the AstraZeneca trials to observe precise levels of protection in this group, but data on immune responses were very reassuring."
Mr Johnson said he was not concerned by Germany's recommendation, adding that the UK's watchdog, the Medicines and Healthcare products Regulatory Agency (MHRA), had "made it very clear" that the AstraZeneca vaccine is "very good and efficacious" and gives a "high degree of protection after just one dose, and even more after two doses".
Speaking during a visit to Scotland, the prime minister added: "The evidence that they've supplied is that they think it is effective across all age groups [and] provides a good immune response across all age groups, so I don't agree with that [Germany's recommendation]."
An AstraZeneca spokesperson said the latest analyses of clinical trial data for its vaccine "support efficacy in the over 65 years age group", adding that the firm was awaiting "a regulatory decision on the vaccine by the EMA in the coming days".


All of the regulators and experts in different countries have been looking at the same data on the Oxford-AstraZeneca vaccine.
That data comes from clinical trials, and those did recruit fewer elderly people overall.
That's because they started off first with younger volunteers to get results as quickly as possible, given the urgency to find out if a vaccine would work well enough to help get us out of the pandemic.
The scientists who ran the trials have always been upfront about this.
But they say there is other evidence to suggest the vaccine will work well in older adults.
Studies show the over-65s have a strong immune responses to the vaccine. After receiving the shots their blood has plenty of the required antibodies that can fight coronavirus.
The UK has been using the AstraZeneca vaccine in its mass immunisation programme for weeks now and should soon have more proof from the real world setting about how much protection the shots give.

Prof Paul Hunter, of the University of East Anglia, told BBC News that the elderly should not worry about receiving the jab.
He said: "I'm almost 65 myself and I would happily take any of the vaccines, including the Oxford-AstraZeneca vaccine. We do know that it is safe in people over 65.
"They have much fewer side effects than younger people and it almost certainly provides substantial benefits in terms of preventing severe disease and reducing the chances of going into hospital."
'No concerns' over safety
Prof Adam Finn, a member of the Joint Committee on Vaccination and Immunisation (JCVI), told BBC's Radio 4's PM programme that Germany's recommendation "just reflects different levels of caution", adding: "What they are basically doing is saying 'We'd like to wait a big longer and know a bit more before we move."
Prof Finn, professor of paediatrics at the University of Bristol, said: "We have no concerns about the safety of the vaccine in any age group".
Dr Doug Brown, chief executive of the British Society for Immunology, said governments both in the UK and elsewhere had to "make important policy decisions in the face of fast-moving situations where our knowledge of the underlying science is still evolving" and that it should not be surprising when it sometimes results in governments "making different decisions from each other".
He added that the MHRA would have "carefully scrutinised the evidence" on the AstraZeneca jab before making their recommendation, and referred to its original report which stated that there is "there is limited information available on efficacy in participants aged 65 or over, although there is nothing to suggest lack of protection".
He said this reflected a "need for more data to be gathered and published on the effectiveness of the vaccine in this age group", but that the jab had shown a "good safety profile" in all published reports.
Meanwhile, the EU and AstraZeneca are involved in a row over vaccine supply shortages.
AstraZeneca has previously said it could deliver only a fraction of the doses between January and March that it had promised to the bloc, blaming production issues at EU plants for a reported 50 million-dose shortfall.
The EU has demanded that UK-made jabs are diverted to mainland Europe to fulfil contractual obligations.
But Cabinet Office minister Michael Gove has said there would "be no interruption" to UK vaccine supplies.
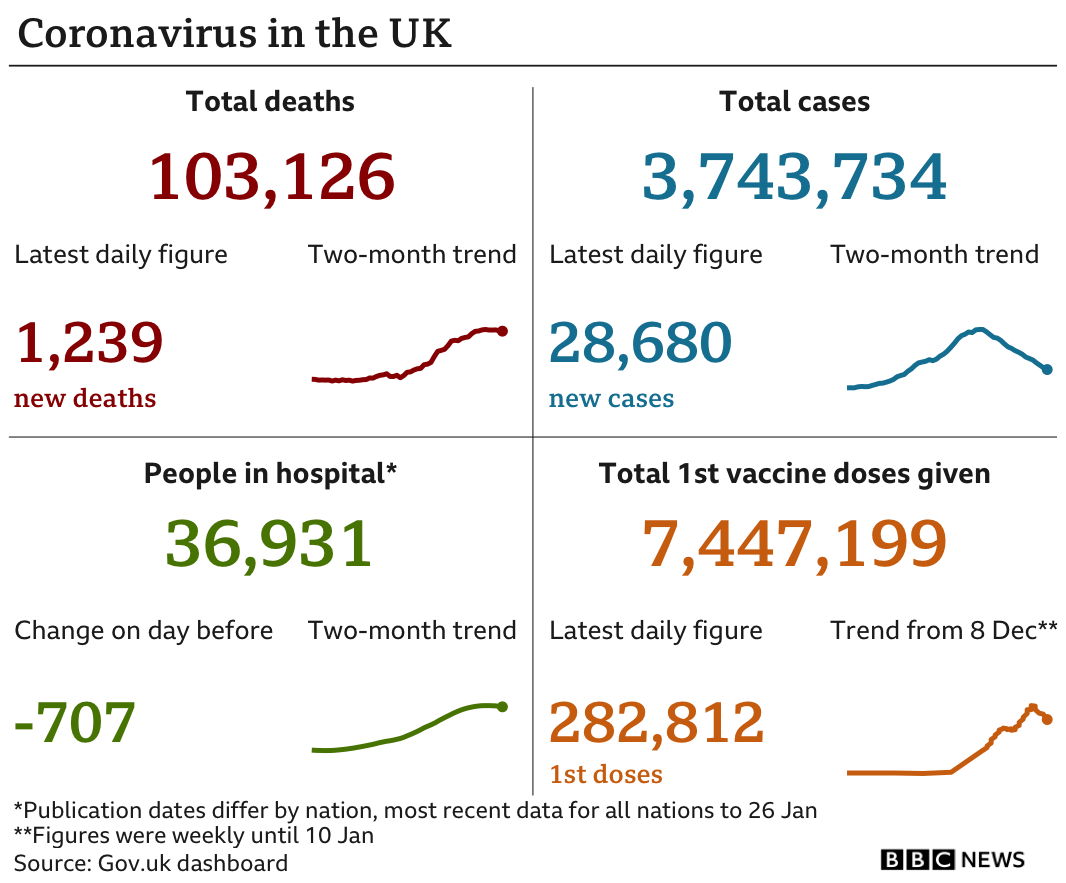
It comes as the UK recorded a further 1,239 deaths within 28 days of a positive coronavirus test on Thursday, according to government figures. There have also been another 28,680 new infections.

- LOCKDOWN: The rules in your area
- TESTING: How do I get a virus test?
- LOOK-UP TOOL: How many cases in your area?
- TRAVEL: What are the UK's rules?

In other developments:
- Scotland is "on track" to meet its Covid-19 vaccine targets, First Minister Nicola Sturgeon has said
- One in three police officers has been threatened by someone who they believed had coronavirus, a survey suggests
- Scientists behind a study tracking coronavirus in England say there are signs of a "shallow decline" in infection levels
- Welsh Secretary Simon Hart says a UK-wide policy for reopening schools would make sense
- Black MPs from across the parties have joined together in a video encouraging people to get a Covid vaccine


Have you been affected by vaccine supply issues? Share your experiences by emailing haveyoursay@bbc.co.uk.
Please include a contact number if you are willing to speak to a BBC journalist. You can also get in touch in the following ways:
- WhatsApp: +44 7756 165803
- Tweet: @BBC_HaveYourSay
- Upload pictures or video
- Please read our terms & conditions and privacy policy
If you are reading this page and can't see the form you will need to visit the mobile version of the BBC website to submit your question or comment or you can email us at HaveYourSay@bbc.co.uk. Please include your name, age and location with any submission.
https://news.google.com/__i/rss/rd/articles/CBMiJmh0dHBzOi8vd3d3LmJiYy5jby51ay9uZXdzL3VrLTU1ODQ3Mzg30gEqaHR0cHM6Ly93d3cuYmJjLmNvLnVrL25ld3MvYW1wL3VrLTU1ODQ3Mzg3?oc=5
2021-01-28 18:51:00Z
52781336752031