The Oxford-AstraZeneca vaccine has been approved for use in the UK, with the first doses due to be given on Monday amid rising coronavirus cases.
The UK has ordered 100 million doses - enough to vaccinate 50 million people.
This will cover the entire population, when combined with the full order of the Pfizer-BioNTech jab, Health Secretary Matt Hancock said.
It comes as millions more people in England are expected to be placed under the toughest tier four restrictions.
On Tuesday, 53,135 new Covid cases were recorded in the UK - the highest single day rise since mass testing began - as well as 414 more deaths within 28 days of a positive test.
Prime Minister Boris Johnson called the vaccine development "a triumph" for British science, adding: "We will now move to vaccinate as many people as quickly as possible."
And England's chief medical officer Chris Whitty praised the "considerable collective effort that has brought us to this point".
Speaking on BBC Breakfast, Mr Hancock said it marked a "significant moment" in the fight against the virus, adding that "2021 can be a year of hope and recovery because we can see our way out of the pandemic".
The Oxford-AstraZeneca vaccine was designed in the first months of 2020, tested on the first volunteer in April, and has since been through large-scale clinical trials involving thousands of people.
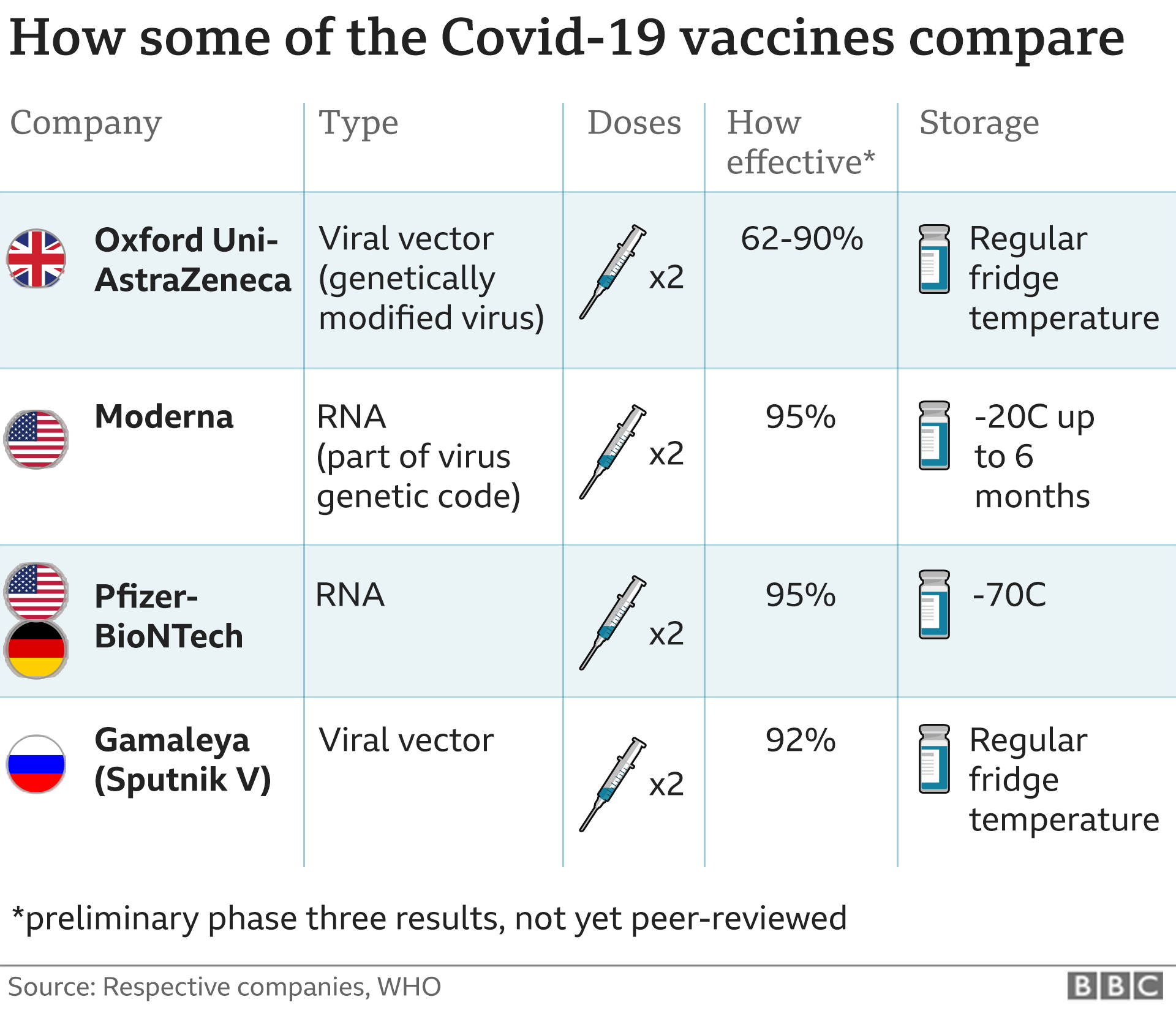
It is the second jab to be approved in the UK after the Pfizer-BioNTech vaccine was given the go-ahead in December.
Unlike the Pfizer-BioNTech jab, which needs ultra cold storage at -70C, the Oxford-AstraZeneca can be stored in a standard fridge so will be easier to deliver to care homes and GP surgeries.
AstraZeneca's chief executive Pascal Soriot told BBC Radio 4's Today programme the company will "progressively ramp up" the vaccination programme, and will be able to deliver up to two million doses a week.
More than 600,000 people in the UK have been vaccinated since Margaret Keenan became the first in the world to be given it outside of a clinical trial.


The approval of the Oxford-AstraZeneca vaccine marks a major turning point in the pandemic.
It will lead to a massive expansion in the UK immunisation campaign aimed at getting life back to normal.
The shift to giving as many people as possible the first doses of either approved vaccine effectively doubles the number of people given some protection.
The second dose, which gives maximum protection, will come up to three months later.
However, the next couple of months still look bleak.
Health officials have spoken of "unprecedented" levels of infection and some hospitals are struggling with the number of patients.
The restrictions we are all living under are likely to get tougher before vaccines make a tangible difference and sets us on the path to normality.

Prof Andrew Pollard, director of the Oxford Vaccine Group, told BBC Radio 4's Today programme the vaccine approval was an "astonishing achievement" in science and clinical research.
But he said there was still "more work to do", warning: "It's not over yet."
"Our colleagues in hospital are facing some real horrors caused by this virus. The next steps are critical," he said.
Meanwhile, the health secretary has said that more areas will be placed under England's toughest tier four - "stay at home" - restrictions.
Mr Hancock will set out further details of the changes in the House of Commons later.
He has urged people to "stay in this new year" regardless of what tier their local area is under - in a bid to slow the spread of the virus.
Education Secretary Gavin Williamson will also be making a statement later about the return of schools in England in January, Mr Hancock said.
The new vaccine approval comes after Public Health England said the country was facing "unprecedented" levels of infections, and health officials in parts of Wales, Scotland and the south of England voiced concerns about the increasing pressure on the NHS.

Who will get the vaccine?
Priority groups for immunisation - including the elderly, care home residents and health and care workers - have already been identified.
The immunisation campaign will now shift to giving as many people as possible their first dose of vaccine.
The aim will be to give as many vulnerable people some protection from Covid-19.
The decision is based on advice from the Joint Committee on Vaccination and Immunisation.
A Department of Health and Social Care spokesperson said: "The priority should be to give as many people in at-risk groups their first dose, rather than providing the required two doses in as short a time as possible.
"Everyone will still receive their second dose and this will be within 12 weeks of their first. The second dose completes the course and is important for longer term protection."
How effective is the Oxford vaccine?
There are three figures doing the rounds - 62%, 70% and 90%.
The first analysis of the trial data showed 70% of people were protected from developing Covid-19 and nobody developed severe disease or needed hospital treatment.
The figure was just 62% when people were given two full doses of the jab and 90% when they were first given a half dose and then a full one.
The Medicines and Healthcare products Regulatory Agency (MHRA) has approved two full doses of the Oxford-AstraZeneca vaccine.
However, unpublished data suggests that leaving a longer gap between the first and second doses increases the overall effectiveness of the jab.
There was not enough clear data to approve the half-dose, full-dose idea.
All the vaccines are expected to be equally effective against the new variants of the virus that have emerged.
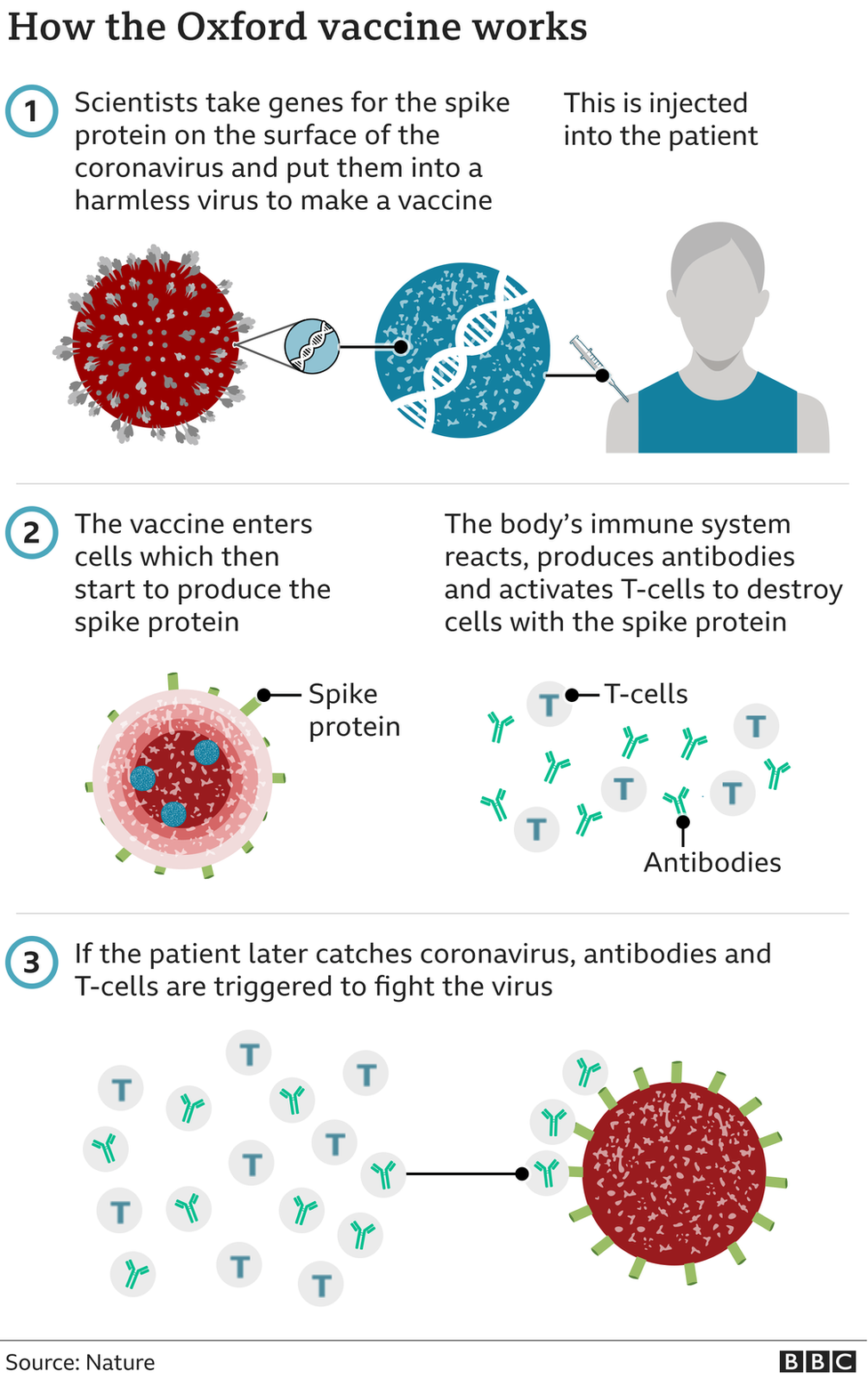
How does it work?
The vaccine is a genetically modified common cold virus that used to infect chimpanzees.
It has been altered to stop it causing an infection in people and to carry the blueprints for part of the coronavirus, known as the spike protein.
Once these blueprints are inside the body they start producing the coronavirus' spike protein, which the immune system recognizes as a threat and tries to squash it.
Then, when the immune system comes into contact with the virus for real, it already knows what to do.
Follow James on Twitter.

Do you have any questions about the coronavirus vaccine? Email haveyoursay@bbc.co.uk.
- WhatsApp: +44 7756 165803
- Tweet: @BBC_HaveYourSay
- Please read our terms & conditions and privacy policy
Or use this form to get in touch:
If you are reading this page and can't see the form you will need to visit the mobile version of the BBC website to submit your comment or send it via email to HaveYourSay@bbc.co.uk. Please include your name, age and location with any comment you send in.

https://news.google.com/__i/rss/rd/articles/CBMiKmh0dHBzOi8vd3d3LmJiYy5jby51ay9uZXdzL2hlYWx0aC01NTI4MDY3MdIBLmh0dHBzOi8vd3d3LmJiYy5jby51ay9uZXdzL2FtcC9oZWFsdGgtNTUyODA2NzE?oc=5
2020-12-30 09:15:00Z
52781275240124






